
It is always positive because the removal of an electron always requires that energy be put in (i.e., it is endothermic). 
IE is usually expressed in kJ/mol of atoms. is the amount of energy required to remove an electron from an atom in the gas phase: A(g) → A + (g) + e − Δ H ≡ IE Ionization energy (IE) The amount of energy required to remove an electron from an atom in the gas phase. Referring only to a periodic table and not to Figure 8.19 "Atomic Radii Trends on the Periodic Table", which atom is smaller, Ca or Br? S is above Te on the periodic table, so Te is larger because as you go down the column, the atoms get larger.Si is to the left of S on the periodic table, so it is larger because as you go across the row, the atoms get smaller.Referring only to a periodic table and not to Figure 8.19 "Atomic Radii Trends on the Periodic Table", which atom is larger in each pair? Again, we can summarize this trend as follows: as → PT, atomic radius ↓įigure 8.19 "Atomic Radii Trends on the Periodic Table" shows spheres representing the atoms of the s and p blocks from the periodic table to scale, showing the two trends for the atomic radius. 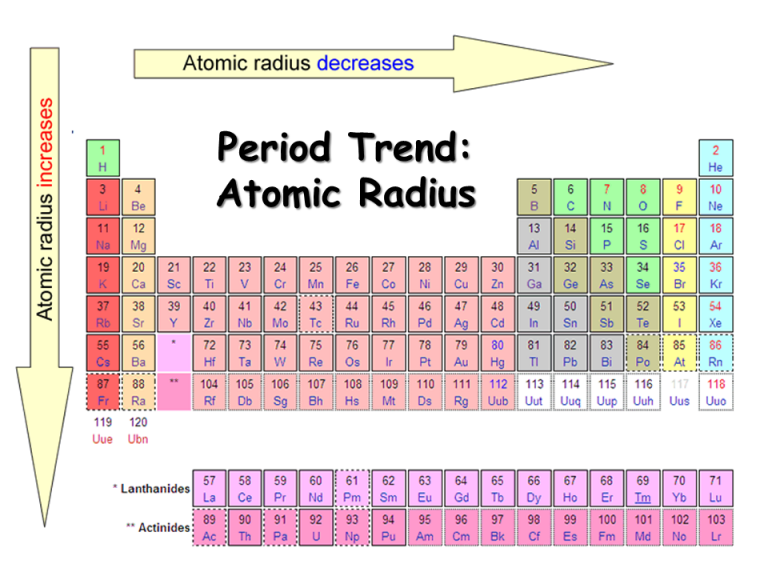
The increasing positive charge casts a tighter grip on the valence electrons, so as you go across the periodic table, the atomic radii decrease. This is because although the valence shell maintains the same principal quantum number, the number of protons-and hence the nuclear charge-is increasing as you go across the row. Going across a row on the periodic table, left to right, the trend is different. This trend can be summarized as follows: as ↓ PT, atomic radius ↑ This is because the valence electron shell is getting a larger and there is a larger principal quantum number, so the valence shell lies physically farther away from the nucleus. Such radii can be estimated from various experimental techniques, such as the x-ray crystallography of crystals.Īs you go down a column of the periodic table, the atomic radii increase. Although the concept of a definite radius of an atom is a bit fuzzy, atoms behave as if they have a certain radius. The atomic radius An indication of the size of the atom. The first periodic trend we will consider atomic radius. There may be a few points where an opposite trend is seen, but there is an overall trend when considered across a whole row or down a whole column of the periodic table. There is no other tool in science that allows us to judge relative properties of a class of objects like this, which makes the periodic table a very useful tool. 
The variation of properties versus position on the periodic table is called periodic trends Variation of properties versus position on the periodic table. One of the reasons the periodic table is so useful is because its structure allows us to qualitatively determine how some properties of the elements vary versus their position on the periodic table. Be able to state how certain properties of atoms vary based on their relative position on the periodic table.More detailed molar volume data for the elements can also be found on another page at the same website. Obviously, their molar volume would be higher than that of the transition metals that come before them as those are significantly more dense.ĭensity data for the elements can be found here. The density values for the non-metals seem to display some variability so I am not sure how the their molar volume trend can be explained. However, once we reach the non-metals, the trend seems to reverse. Mass density generally increases from group 1 to group 13 resulting in the fall in molar volume across the period initially while for the transition metals, they stay roughly constant and take relatively large values. As such, they are seen as spikes in the graph, with the highest molar volumes in their respective periods. The molar masses of the group 1 metals are considerably large but yet they have a relatively low mass density. With this understanding, it is easier to explain the trend that you have provided. As such, molar volume would not only be dependent on atomic radius but would also depend on the structure and bonding of the atoms. Molar volume is a macroscopic property that is defined by Wikipedia as the ratio of the molar mass to the mass density of the substance at a certain temperature and pressure. Typically, one would understand "atomic volume" to be the volume of an atom of the element and it is directly related to the radius of the atom. Firstly, we need to clearly define "atomic volume" and "molar volume".
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
